- Home
- Resources
- Phenotyping
- PhenoSense
Monogram Biosciences

PhenoSense®
PhenoSense®, offered as a single HIV drug resistance test and in the combination test PhenoSense® GT, represents cutting-edge resistance testing technology. PhenoSense provides a direct, quantitative measurement of how well a patient's virus responds to antivirals. PhenoSense drug resistance results are provided with a measure of viral fitness or replication capacity.
PhenoSense drug resistance results are provided with a measure of viral fitness represented by replication capacity.
PhenoSense utilizes advanced molecular biology and cellular virology techniques to determine a rapid and precise measure of susceptibility to antiretroviral drugs. The capacity to perform these techniques in a reliable and robust, high-throughput manner is a core strength of Monogram Biosciences.
The Technical Process: Step by Step
1. Isolating the viral RNA
- Protease (PR) and reverse transcriptase (RT) sequences of the patient’s HIV virus are isolated from the patient's blood sample and amplified via reverse transcription/polymerase chain reaction (RT-PCR).
- The process is optimized and validated to capture sequences representing the viral diversity present in the patient sample.
2. Constructing the test vector
- Patient HIV PR and RT sequences are inserted into the test vector (see figure below).
- Luciferase, a light-emitting reporter gene, is present in the test vector.
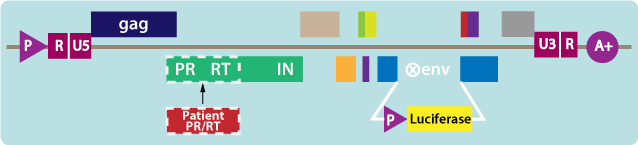
3. Producing and testing the virus
- The test vector is introduced into host cells to produce virus particles that incorporate the PR and RT from the patient’s HIV.
- These virus particles are dependent on the activity of the patient’s virus PR and RT to replicate and are used to infect target cells in the presence and absence of specific antiretroviral drugs.
- The presence of luciferase glow in the target cells indicates the ability of the virus to grow at increasing levels of drug concentration. The concentration of drug that can inhibit the growth of the virus is a measure of the susceptibility of the virus to the different drugs.
4. Measuring resistance
- The drug susceptibility of the patient-derived test virus is compared to that of a reference virus that is susceptible to all of the drugs tested.
- Susceptibility of patient virus is expressed as the fold change†, and the fold change is compared to cutoff values determined through correlation of drug susceptibility and clinical outcomes data.
†Fold change: A ratio of the drug concentration needed to inhibit the patient’s virus relative to that of a wild-type control strain.

